Delivery Platform

MilanaPharm’s patented drug delivery platform, BioGel XR, consists of a combination of a tri block copolymer and a natural polysaccharide. The system is designed to take advantage of body temperature to undergo sol-to-gel transition and can be presented, to name a few, as a liquid (viscous or dilute), a semi-solid (gel/paste), a spray, or a foam to suite the need/purpose. In addition, a number of different drugs can be incorporated to treat various conditions. BioGel XR meets the biocompatibility testing requirements of ISO/USP.
Research has shown that drug delivery is key to optimizing drug therapy. MilanaPharm’s hydrogel based technology is an innovative platform designed to optimize drug therapy. Our platform, BioGel XR, focuses on three logics/features of in-situgelation, sustained-release and bioresorption and adhesion. The three logics, when combined provide ease of use, better tolerability and therapy compliance.
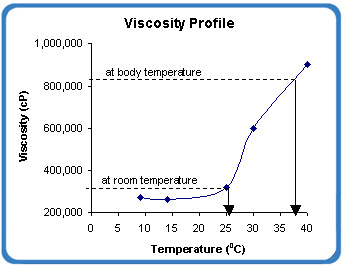
Fig 1: Viscosity profile of BioGel XR
In-situ gelation
MilanaPharm’s proprietary in-situ gel system, BioGel XR, is a viscous liquid and undergoes solution to gel (sol-to-gel) transition using physiological condition such as body temperature as the trigger. This property allows the Company’s in-situ gel platform to (a) be easily and directly placed on or at the site of action and (b) transform to a gel following application.
By the process of “reverse thermal gelation”, the viscosity of BioGel XR increases 2 – 4x of its viscosity at room temperature (Fig 1).
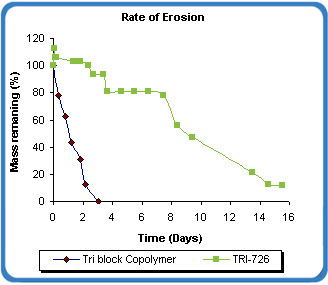
Fig 2: Erosion rate of BioGel XR matrix
Sustained-erosion
The platform has the ability to erode over a period of several hours to several days. This feature not only allows for extended residence of the matrix but also for slow release of the incorporated drug over this period.
In the example on the left, the erosion rate of the BioGel XR matrix is measured as an estimate of the duration of matrix residence time. The matrix erodes over 15 days under controlled condition, which represents a 5-fold increase over the tri block copolymer alone (Fig 2).
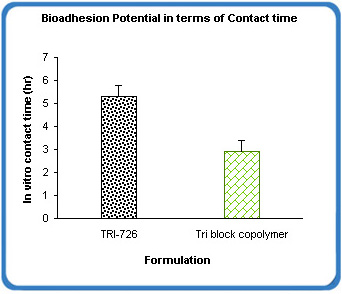
Fig 3: Bioadhesion potential of BioGel XR matrix
Bioresorption and adhesion
BioGel XR uses ingredients that are biocompatible with no known adverse reactions. The ingredients are hydrophilic as well, making the formulation slowly dissolve in body fluids (bioresorb) and body water (intra and extracellular). This logic provides the advantage of not requiring subsequent removal at the end of a treatment period. In addition, the platform exhibits excellent ability to bioadhere at the site of application.
BioGel XR showed excellent bioadhesion potential measured in terms of contact time (the time for which a steel disc of certain weight impregnated with the formulation adhered to a glass plate coated with mucin that was placed over a water bath maintained at 37° C). The contact time was approximately 2x that measured with the tri block copolymer alone (Fig 3).

